Progenitor cells offer great hope for heart failure patients
The 5D Heart Patch Project, led by Prof Kenneth Chien, has identified human ventricular progenitor (HVP) cells that can create self-assembling heart grafts in vivo. The research has the potential to offer hope to millions of people suffering from heart failure.

Following injury, the human heart has a very limited capacity to regenerate contracting cardiac muscle, leading to heart failure, a leading cause of morbidity and mortality worldwide. The only cure for the millions of patients with end-stage heart failure is heart transplantation, which is severely limited by the few thousand available donor hearts each year.
An ERC Advanced Grant supported the 5D-Heart Patch program with the ambitious goal of unlocking the therapeutic potential of generating billions of human heart precursors or “progenitors” from human embryonic stem cells by forming muscle graft patches in the intact heart. The program has reached its initial goals and is now completed.
Over the past 5 years, a team of physicians and scientists in the Department of Cell and Molecular Biology and the Department of Medicine Huddinge, along with partners in the academic as Technical University of Munich (TUM) and in the private sector AstraZeneca and Procella, have moved this technology to improve the function of the pig heart following myocardial injury by the generating 3-D heart graft patches that have the added advantages of durability and integration with the native heart, i.e., a 5-D heart patch.
At AstraZeneca, the work has progressed to clinical development, and is projected for human studies in the next few years.
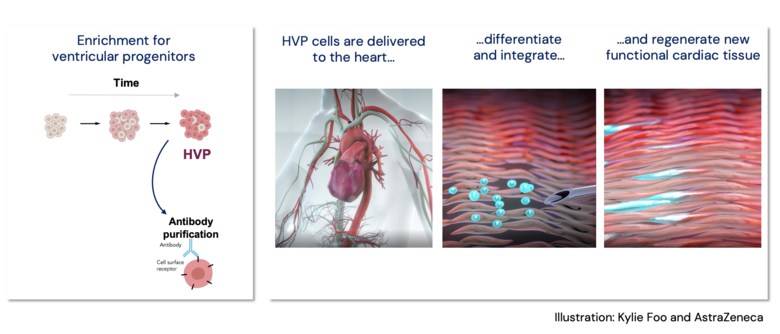
The ability to generate heart muscle cells from embryonic stem cells has been known for decades, but the ability to generate an in vivo heart muscle graft patch has been limited by a) the need to locate the graft near the area of injury, b) to develop adequate blood flow as the patch enlarges to form a 3-D structure, and c) the consequent fibrosis that forms around transplanted cardiac muscle cells.
The current study shows that human embryonic stem cells can be directed to form billions of heart muscle progenitors that have the intrinsic property of migrating to the site of heart injury, subsequently expanding to a 3-D structure, stimulating the formation of blood vessels, and also preventing fibrosis that would interfere with their communication with the native heart muscle (which is key to avoid life threatening arrythmias and maintaining an organized heart rhythm). These properties are not found in already contracting heart muscle cells and suggests that these progenitor cells could be an ideal source for generating a 5-D heart muscle graft. The studies set a solid scientific foundation for a novel approach to achieve heart muscle regeneration, giving hope to patients with end-stage heart failure who are waiting for a heart transplant.
Publications
Migratory and anti-fibrotic programmes define the regenerative potential of human cardiac progenitors.
Poch CM, Foo KS, De Angelis MT, Jennbacken K, Santamaria G, Bähr A, Wang QD, Reiter F, Hornaschewitz N, Zawada D, Bozoglu T, My I, Meier A, Dorn T, Hege S, Lehtinen ML, Tsoi YL, Hovdal D, Hyllner J, Schwarz S, Sudhop S, Jurisch V, Sini M, Fellows MD, Cummings M, Clarke J, Baptista R, Eroglu E, Wolf E, Klymiuk N, Lu K, Tomasi R, Dendorfer A, Gaspari M, Parrotta E, Cuda G, Krane M, Sinnecker D, Hoppmann P, Kupatt C, Fritsche-Danielson R, Moretti A, Chien KR, Laugwitz KL
Nat Cell Biol 2022 May;24(5):659-671
Related News & Views:
New cell therapy approach to regenerate cardiac tissue following a heart attack
Defining the pathways of heart regeneration.
Timmer LT, van Rooij E
Nat Cell Biol 2022 May;24(5):606-607
