Volker Lauschke has been awarded the Swedish Research Council's Consolidation Grant
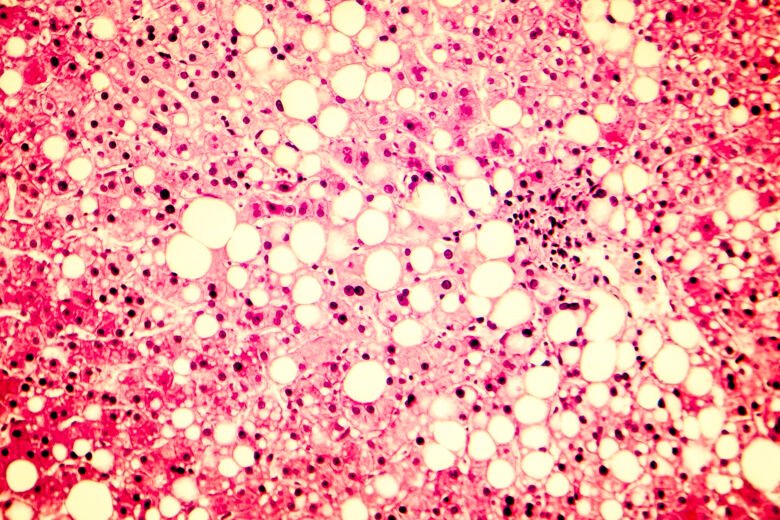
KI researcher Volker Lauschke has received the Swedish Research Council's Consolidation Grant for the research project "Modulation of Tissue Communication to Combat Non-Alcoholic Fatty Liver Disease" regarding the severe liver disease NASH. The Swedish Research Council's Consolidation Grant amounts to 7.4 million SEK over five years. This marks the fifth time Volker Lauschke has been awarded funding from the Swedish Research Council.
What is the overall goal of your research?

"Non-alcoholic steatohepatitis (NASH) is a common liver disease affecting up to 2-6 percent of the general population and 15-40 percent of overweight individuals. NASH is characterized by fatty liver, chronic inflammation, and damage to liver cells and has the potential to progress to liver cirrhosis and liver cancer. Despite extensive efforts, there is currently no approved treatment for NASH," says Volker Lauschke , professor at the Department of Physiology and Pharmacology, Karolinska Institutet.
"In the research project, we will integrate ex vivo tissue models from the liver, pancreas, skeletal muscle, and fat to map interactions in metabolic diseases and identify new endocrine signals contributing to NASH etiology and progression."
"We will also use the platform to screen chemogenomic libraries and identify compounds that activate healthy signals and inhibit disease signals. The project thus provides a new perspective on NASH as a complex pathology caused by dysregulated tissue interactions and its disease mechanisms, which are overlooked by current drug development programs, ultimately aiming to develop effective treatments."
How will the grant be used?
"The Consolidation Grant will support progress in three areas. Firstly, for the further development of organotypic and microphysiological primary human tissue models. Secondly, for comprehensive mapping of tissue-specific molecular signatures in metabolic health and disease. And thirdly, for chemical screening to develop drug candidates against identified targets."
