Vulnerable human spinal motor neurons can be rescued from degeneration
Researchers at Karolinska Institutet reveal mechanisms of neuron vulnerability and resistance in spinal muscular atrophy (SMA) by investigating gene activity dynamics of different neurons in a mouse model. The study was recently published in the journal Genome Research.

In the disease spinal muscular atrophy (SMA), which mainly affects young children under the age of 2, motor neurons, which control all skeletal muscles in the body, degenerate. However, there are certain groups of motor neurons that, for unknown reasons, are selectively resistant, e.g. the ocular motor neurons that innervate (control) the eye muscles.
“Spinal muscular atrophy is caused by a loss of a "survival motor neuron protein 1" (SMN1). The SMN1 protein is found in all cells in the body and is among other things necessary for the metabolism of ribonucleic acid (RNA), the messenger molecules of our genome”, says Eva Hedlund, group leader at the Department of Neuroscience.
Why does the loss of this protein hit the motor neuron so hard?
“Genes that are active in specific neuronal types define their unique identities and functions in health as well as their susceptibility to various degenerative diseases” Eva Hedlund continues.
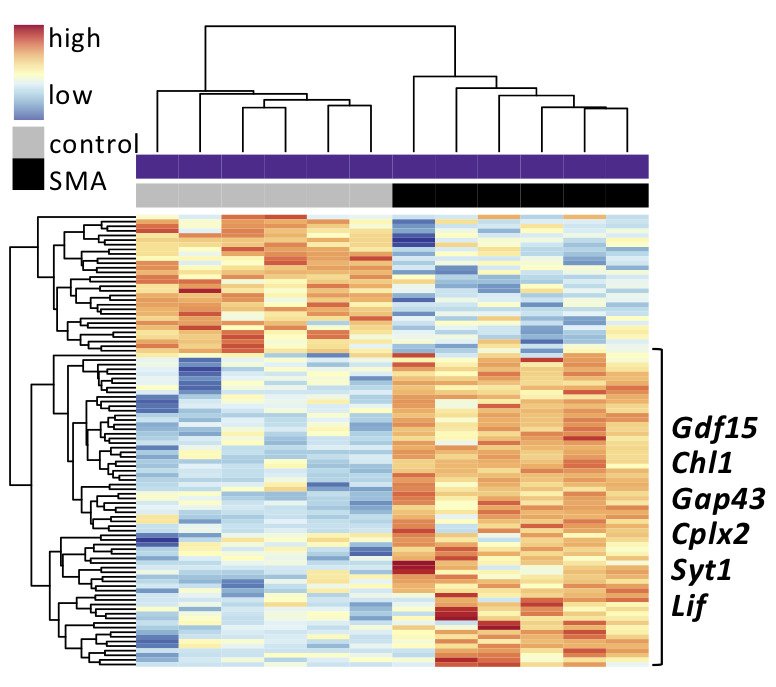
“Factors within the motor neuron itself seem to be decisive for their sensitivity in SMA, which has led us to assume that inherent properties also affect the resilience of certain neurons”, says Susanne Nichterwitz, previously PhD student in the lab and first author of the study, and now postdoc at UCLA, CA.
To reveal mechanisms of neuron vulnerability and resistance in SMA, the researchers examined the dynamics of gene activity in muliple different neuron types (sensitive and resistant) in a mouse model of SMA by studying messenger RNAs which show which genes are active.

What are the most important results?
"Among other things, our analysis shows that all neurons have a unique response to the loss of SMN1. Furthermore, at each individual point in the disease, the neuron demonstrates a unique transcriptional response, which means that in order to understand the disease and cell’s adaptation to it, cells need to be studied longitudinally," says Eva Hedlund.
"We were also able to show that ocular motor neurons demonstrate unique adaptation mechanisms during the course of the disease that may explain their resilience," Susanne continues.
Recently, the first treatments for SMA were approved, aimed at SMN, with very promising results. But the beneficial effects vary depending on the time of treatment and the severity of the disease.
Patients are likely to benefit from additional complementary therapies aimed at maintaining or improving motor function.
“We hope that the factors we have identified in ocular motor neurons, including Gdf15, which appear to act as a defence against the disease, can be used as complementary therapies for patients to preserve sensitive motor neurons”, Eva Hedlund concludes.
The study was financed with grants from the Swedish Research Council, the European Union Joint Programme for Neurodegenerative Disease (JPND), the Swish National Science Foundation and Karolinska Institutet.
Publication
"LCM-seq reveals unique transcriptional adaptation mechanisms of resistant neurons and identifies protective pathways in spinal muscular atrophy".
Nichterwitz S, Nijssen J, Storvall H, Schweingruber C, Comley LH, Allodi I, van der Lee M, Deng Q, Sandberg R, Hedlund E
Genome Research, Published in Advance August 20, 2020, doi:10.1101/gr.265017.120
