Ongoing study is testing the COVID-19 vaccine on patients with compromised immune systems
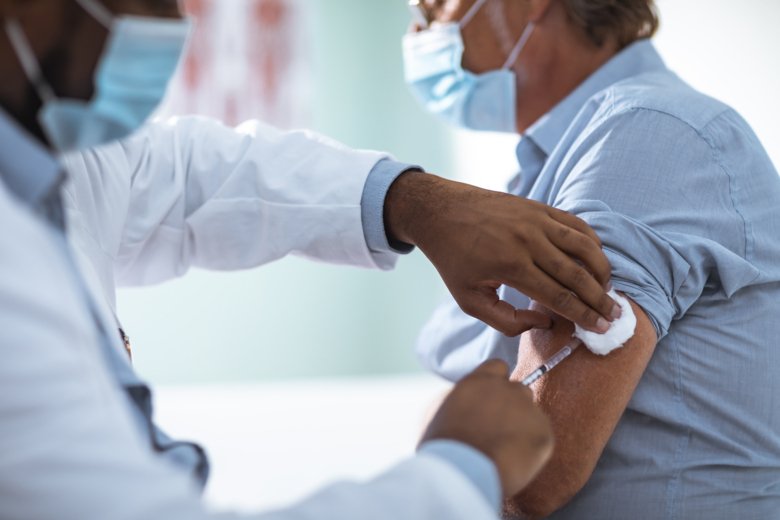
Researchers at Karolinska Institutet are investigating the efficacy and safety of COVID-19 vaccines in a new study on patients with compromised immune systems, who can become seriously ill if they are affected by COVID-19. On February 23, the first patient in the study was vaccinated with an mRNA vaccine at Karolinska University Hospital. So far, the project has received two grants from the Knut and Alice Wallenberg Foundation of SEK 10 million in total.
The clinical study is a joint effort between different departments at Karolinska University Hospital and several research groups at Karolinska Institutet and SciLifeLab. Patients with compromised immune systems will receive an mRNA vaccine against COVID-19, after which the vaccine response will be studied, and any side effects followed up. In addition, detailed analyses of different parts of the immune system will be carried out using blood, saliva and faeces samples.
Poorly studied

Patients with compromised immune systems are in many cases very sensitive to COVID-19 disease and mortality rates are higher in this group.
“Efficacy and safety following a COVID-19 vaccination are poorly studied in these patients, and it is therefore very important that we acquire more knowledge,” comments Soo Aleman, senior physician and section manager at the Medical Unit Infectious Diseases at Karolinska University Hospital, associate professor at the Department of Medicine, Huddinge, Karolinska Institutet and co-ordinating investigator for the entire study.
A vaccine stimulates the immune system to build up a defence against microorganisms such as bacteria or viruses, but if the immune system is impaired, there is no guarantee that adequate protection can be formed. There are today many categories of patients living with compromised immune systems.
Evidence-based information

Hans-Gustaf Ljunggren, professor at the Center for Infectious Medicine, Department of Medicine, Huddinge, Karolinska Institutet is in charge of the organisation that receives all samples and contributes to analyses.
“Knowledge of vaccine responses is important in this group, as healthcare must be able to provide evidence-based information about vaccine effects during the current pandemic,” says Hans-Gustaf Ljunggren. “It is also important to create new knowledge about the conditions necessary to build up a protective immune response following vaccination against COVID-19.”

Margaret Sällberg Chen, professor at the Department of Dental Medicine at Karolinska Institutet, is in charge of one of several sub-projects within the study. Her research group has just completed a study on saliva immunity, where they have established a new very sensitive method for measuring antibodies in saliva together with researchers at KTH. The method will now be used for the new study where the researchers will follow antibody responses in saliva in vaccinated risk groups and healthy controls.
Grants from KAW
The study is approved by the Medical Products Agency and the Ethical Review Authority. So far, the project has received two grants from the Knut and Alice Wallenberg Foundation (KAW) via SciLifeLab. SEK 5 million has been granted to Soo Aleman as the main applicant as part-funding for the clinical trial. Another SEK 5 million has been allocated to professor Karin Loré, an expert in vaccination immunology who has studied mRNA vaccine for several years and has been involved in testing new COVID-19 vaccines at the Department of Medicine, Solna, Karolinska Institutet, and Stephan Mielke, director of the Centre for Allogeneic Stem Cell Transplantation and Cell Therapy (CAST) at Karolinska University Hospital and professor at the Department of Laboratory Medicine at Karolinska Institutet, for the sub-study conducted at CAST.

“There is still very little knowledge about how individuals with, for example, absent or very few B cells respond to COVID-19 vaccination and whether a modified vaccination strategy may be needed to achieve adequate protection,” says Karin Loré. “We plan to make in-depth analyses of the quality and durability of the vaccine response in this patient group compared with healthy controls. A project like this would not be possible without close collaboration between clinicians and researchers with different expertise.”
Financial support has also been received from the patient associations Blodcancerförbundet (the Blood Cancer Association) and Primär Immunbrist Organisationen (the Primary Immunodeficiency Organisation (PIO)). Other research grants are also being used in order to carry out the study.
This news article is partly based on a press release from Karolinska University Hospital, with the addition of new information.
Participating principal researchers at Karolinska Institutet
Principal investigators at Karolinska University Hospital:
Soo Aleman, Department of Medicine, Huddinge
Peter Bergman, Department of Laboratory Medicine
Piotr Nowak, Department of Medicine/Department of Laboratory Medicine, Huddinge
Gunnar Söderdahl, Department of Clinical Science, Intervention and Technology
Stephan Mielke, Department of Laboratory Medicine
Lotta Hansson, Department of Oncology-Pathology
Center for Infectious Medicine, Department of Medicine, Huddinge:
Hans-Gustaf Ljunggren
Marcus Buggert
Department of Medicine, Solna:
Karin Loré
Department of Dental Medicine:
Margaret Sällberg Chen
