New study questions the role of supercomplexes in metabolism and disease
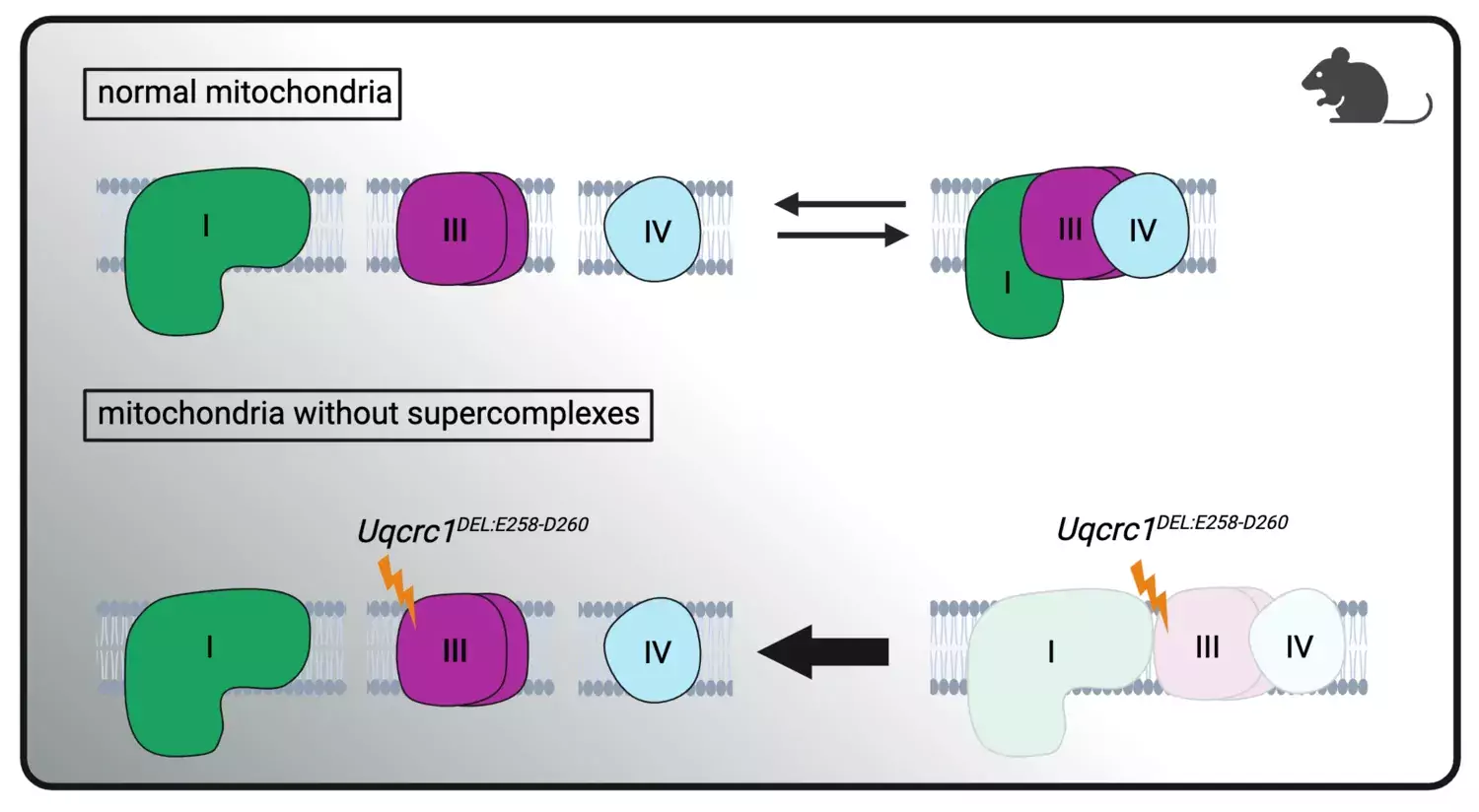
A new study published in Cell Metabolism by the research group of Professor Nils-Göran Larsson, at the Department of Medical Biochemistry and Biophysics, Karolinska Institutet, shows that so-called supercomplexes have no major role in cellular energy production and that altered levels therefore are unlikely to affect physiology and disease.

The food we eat is degraded by metabolism and the energy harvested by mitochondria, which are the powerhouses of our cells. During metabolism, the nutrients we ingest are broken down and electrons are harvested and used by mitochondria to produce the cellular energy currency, called ATP. The production of ATP depends on a very complicated enzyme system called the respiratory chain. The respiratory chain consists of several large protein complexes which in turn can form higher-order complexes, so-called respiratory chain supercomplexes.
“The role of the supercomplexes has been intensively debated for decades and we have addressed this question by creating a very powerful mouse model which contains a normal amount of fully functional respiratory chain complexes, that cannot interact to form supercomplexes. Very surprisingly, these mice are healthy, showing that supercomplex formation is dispensable for normal cellular energy homeostasis”, explains Nils-Göran Larsson, professor at the Molecular Metabolism Division at MBB at Karolinska Institutet.
“Many reports argue that respiratory chain supercomplexes are important in a wide range of physiological processes and human diseases, like diabetes, metabolic disease, heart failure, and cancer. Manipulation of supercomplex formation has been suggested as a new way to treat cancer and metabolic diseases. Our results show that such approaches are unlikely to succeed and that supercomplex formation has no major effect on cellular energy homeostasis”, says Nils-Göran Larsson.
Decreased mitochondrial function is causing a group of inherited diseases that affect children and adults. Decreased mitochondrial function is also an important factor in the progression of common age-associated diseases, such as diabetes, heart failure, and Parkinson´s disease, and contributes to the functional decline seen in aging.
“Despite the huge importance of mitochondrial energy conversion in human disease, many mechanisms are poorly understood. The published results show that supercomplex formation may be dispensable for cellular energy homeostasis and raise the question of alternate ways of regulating energy production”, says Nils-Göran Larsson.
Read the article in Cell Metabolism here
Main financiers of the study
Vetenskapsrådet, KAW, Novo Nordisk Foundation, Diabetesfonden.
Important collaboration partners
Max Planck Society, Cambridge Univ, Utrecht Univ.
