Hearing loss - easy to prevent but hard to cure

One billion young people risk hearing loss in the future if they continue to listen to their headphones at too high a volume. To develop treatments, researchers need to solve the mysteries of the cochlea - well hidden in the inner ear, a pea-sized organ embedded in bone a few centimeters inside the outer ear.
Text: Annika Lund for the magazine Medicinsk Vetenskap issue nr 2, 2023 / Spotlight on hearing loss
What would you find worse - losing your hearing or your sight? This may seem like an impossible question to answer. But one who did is author Helen Keller, who lost both her sight and hearing at the age of about a year and a half after a bout of meningitis. And she described it this way: “Blindness separated me from things while deafness separated me from people.”
However, Helen Keller was born in the last century. Since then, research has fortunately progressed. There is now more knowledge about how both vision and hearing work - and much better help at hand.
How hearing works
In simple terms, hearing works like this: sound waves travel through the ear canal, eardrum and small bones to the inner ear. There they encounter the cochlea which translates the sound waves into something the neurons can pick up. These signals go to the brain where they are interpreted.
Hearing problems caused by problems in the outer or middle ear are common and usually easy to fix. This may involve removing a wax clot or healing a damaged eardrum. Obstructions in these parts of the ear rarely cause permanent hearing problems.
Not so with the cochlea. The conversion from sound wave to activation of neurons is a complex process. When the cochlea deteriorates, we get permanent hearing loss.
And it is common - very common. And it will become even more common.
Globally, around 430 million people have disabling hearing loss. In Sweden, about 1.5 million individuals are affected. More than 560,000 Swedes have hearing aids.
The most common cause of hearing loss is age, followed by noise. As more people reach old age, more people are expected to develop age-related hearing loss.
But the number of noise-related injuries is also expected to increase - significantly. And here our headphone habits throw a nasty spanner in the works.
One billion are at risk of hearing damage
According to a high-profile study, one billion of today's young people (aged 12-34) are expected to suffer from hearing loss as a result of what researchers call “unsafe listening” through headphones. The study, published in 2022 in BMJ Global Health, summarises what is known from other studies about the prevalence of voluntary exposure to excessive volume levels. The researchers then tried to extrapolate the figures to the global level.
The generation surveyed comprises of 2.8 billion individuals. A large proportion of them, or one billion individuals, are at risk of hearing damage - due to the volume level they choose in their headphones.
Facts: Two kinds of hearing measurements
Assessing hearing loss involves investigating both how faint the sounds (the decibel scale) and how high or low the frequency of sounds (the treble scale) you can hear.
About the treble scale: If you roll the cochlea out to an elongated tube, the hair cells along it captures different sounds. Hair cells that are supposed to respond to light or high frequency sounds are closest to the entrance of the cochlea and are therefore usually damaged first.
About the decibel scale: After the age of 40, we lose an average of 10 decibels of hearing ability per decade as we age. This means that a normal conversational tone of 60 decibels will be perceived as 30 decibels after 30 years, which is like a whisper.

“This study was widely disseminated among hearing researchers and sparked debate in many countries. An information campaign is needed here. People do not know enough about volume levels. If a bus goes by, it is perceived as noise, but music at the same volume level is not. But in both cases, hearing is affected," says Barbara Canlon, professor of hearing physiology at the Department of Physiology and Pharmacology at Karolinska Institutet.
The WHO has set some limits on what the ears are considered to tolerate. According to one of these values, sound levels of 90 decibels should reach the ear for no more than 30 minutes per day. This is the same volume level as a lawnmower or a loud restaurant - or the level that many people use in their headphones. For hours and hours, while they are studying or sitting on the bus to work.
However, Barbara Canlon is reluctant to view the WHO limits as a blueprint for safe sound levels.
“There is individual variation. Some people's ears are more sensitive, others are more resilient. So it is more difficult to say what is a safe level for an individual,” she says.
A recommendation from her is to review the settings on your phone. Volume limits can be set on the headphones and alarms can be set for high ambient volume levels. There are also free apps that measure decibel levels.
Barbara Canlon argues that we need to learn to protect our ears from sound exposure - much like “everyone” knows to protect their skin from sun exposure.
“The important message is that this noise-induced hearing loss - it is preventable. It is even simple. Almost everyone has a phone and the apps are free,” she says.
 Photo: Adam Haglund
Photo: Adam HaglundMikael Wiehe about life with hearing loss
Mikael Wiehe's hearing loss affects both himself and those around him. Nowadays, he prefers to perform alone, and restaurant visits only work if the sound environment is right.
She has spent decades doing basic research on hearing and what actually happens when hearing damage occurs. And it seems to work something like this: It is estimated that there are over 16,000 hair cells in the inner ear. Hair cells are activated when they are hit by sound waves, and then they release transmitter substances that are picked up by nerve fibres, the outermost projections of the ear's neurons. Each hair cell communicates with about 20 nerve fibres that together transfer the signals through the neurons to the auditory cortex of the brain.
But hair cells are progressively reduced by natural ageing. They then no longer function properly.
And that is not all. Many nerve fibres also retract. When shortened, they are no longer as accessible to the decreasing amount of neurotransmitters.
This increasing distance between crucial cells in the cochlea is primarily affected by age and noise. But it can also occur as an effect of certain medicines and chemicals, or for other reasons such as infections or blows to the head.
“We have seen in studies on mice that when they are exposed to moderate noise for about two hours, these nerve fibres retract. But they can come back to a certain extent,” says Barbara Canlon.
And when they come back, they are again accessible to neurotransmitters. By then, the sense of hearing has partially recovered from the noise damage.
But if the noise exposure happens again and again and again - repeatedly - then the nerve fibres are unable to recover. They will be permanently shortened. And at the same time, the hair cells may be reduced in number.
Then the hearing loss will be permanent, at least without treatment. Intensive research is underway to find new ways to save hearing.
How researchers are trying to save hearing
One challenge is that the pea-sized cochlea hides its secrets behind a tough shell. It is also located several centimetres inside our hard skull bone - making it more difficult to examine it than the brain itself.
“The cochlea is so inaccessible that studies on human cochlea are rare. This is why virtually all hearing research is done on animals, especially mice. This means that we know less about how human hearing works than we know about human vision, for example,” says Barbara Canlon.
The ongoing global research is still at a fairly basic level, a bit away from having treatments ready. But attempts are being made - and common research goals are to medically enable nerve fibres to pass on signals despite no longer receiving input from hair cells. Or, as Barbara Canlon and her colleagues have done, awaken the hair cells using gene therapy.
They are not the first to use their specific approach; ten years ago it was already used by American researchers who induced hearing in mice with congenital deafness. The mice had a genetic abnormality that prevented them from producing the protein VGLUT3, one of the substances involved in communication between hair cells and nerve fibres. It became global news that the researchers had injected a virus containing the gene for the protein directly into the cochlea, resulting in deaf mice being able to hear. But it is unclear how long the effect lasted. It seems that it was not lifelong.
“This suggests that if this were to develop into a treatment, it would probably need to be repeated at certain intervals. But giving an injection into the cochlea requires a small operation. I find it hard to see people wanting to go through that on a regular basis,” says Barbara Canlon.
She and her colleagues have found another way to get the protein into the cochlea. By injecting it into the back of the head, at a point where it is possible to come into contact with the fluid flowing around and in the brain, the cerebrospinal fluid. That fluid also flows into the so-called cochlear aqueduct, which leads into the cochlea. And researchers have shown that the treatment actually reaches the cochlea through an injection in the back of the head, which is less invasive than an injection directly into the cochlea.
In the previous experiments with injections directly into the cochlea, the mice were newborn, partly because the bones surrounding the cochlea are softer at birth, facilitating the injection. But the Swedish researchers used adult mice. And they also started to hear.
“Promising,” says Barbara Canlon.
“The next step is to conduct experiments on primates. We have contacted a research group that has a group of monkeys of various ages. We want to see if we can get effects on age-related hearing loss. It is possible to imagine that hearing may not be restored, but that the hearing can be partially saved so that a hearing aid will have a better effect,” says Barbara Canlon.

An atlas of cell types in the cochlea
François Lallemend, senior researcher at the Department of Neuroscience at Karolinska Institutet, is also interested in how to rejuvenate the cochlea. But his approach is quite different.
François Lallemend wants to investigate more precisely what actually happens in the cochlea during normal aging. His reasoning is that when hair cells and neurons shut down, a whole range of other cells may have lost function first.
“One could imagine a process that starts with small blood vessels becoming dysfunctional. This could lead to a reduced supply of nerve fibres which then get limited capacity. Or you can imagine that the process starts with changes in various support cells around the hair cells. But these are all just hypotheses that we want to investigate,” he says.
A first goal is to prepare an atlas of the cell types that are actually found inside the cochlea. It is still not fully evaluated.
“We are in a phase where we are collecting cochleas from mice of different ages. So we have not started mapping yet,” says François Lallemend.
The researchers want to use single-cell transcriptomics, a method that makes it possible to see exactly which genes are active in an individual cell. This allows us to understand which molecules the cell produces. And it can describe its functional state and how it interacts with other cells.
This allows you to see if two cells that at first seem very similar, like two neurons, might actually be doing different things. Then perhaps they should be considered as two different cell types. It is already clear that there are several variants of neurons in the ear - but what about the rest of the cell mix - what types of supporting cells, immune cells and connective tissue cells are there? How does the blood supply function, which cells interact with different signaling molecules - and with each other?
“It is possible that each cell type has its own pace of ageing and that they influence each other. We want to investigate this by studying cochleas from mice of different ages and take a systemic approach and investigate the influence of metabolism and immune system with the help of colleagues from other fields” says François Lallemend.
Ultimately, the vision of treating, perhaps mainly delaying, hearing loss with medicinal products is looming.
There are currently no medical treatments for hearing loss. Different types of assistive technology are involved. In addition to hearing aids, there are lots of assistive devices, such as technology that directs sound from the TV or phone directly to the hearing aid.
Cochlear implants are available for all ages
And then there are cochlear implants (CI). They are a solution when regular hearing aids are not enough. Or when someone has gone completely deaf, perhaps after a head injury or meningitis. Or when someone is born deaf.
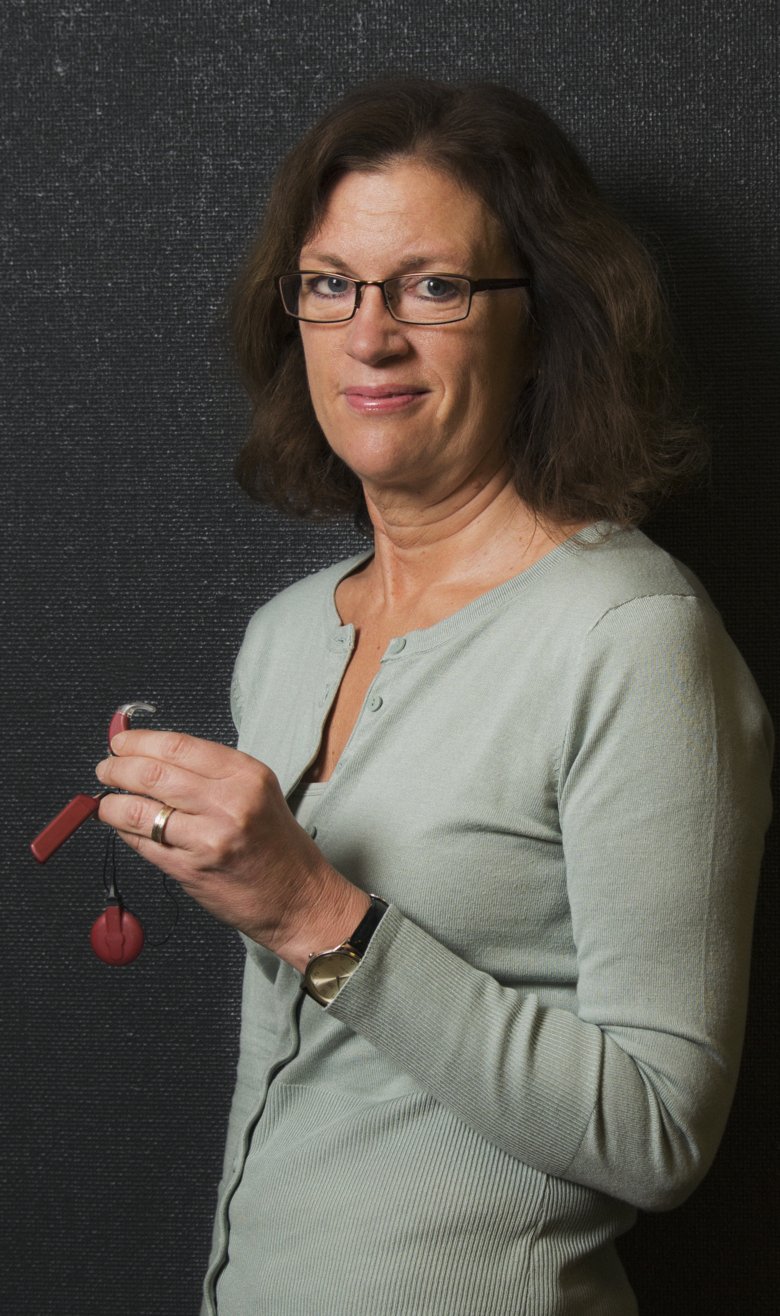
“We have become increasingly generous in assessing who can receive cochlear implants. Nowadays, they are available for people of all ages, from babies to the elderly, with hearing loss and deafness. The oldest person I have operated on was 86 years old, and in her case we found no other cause for the hearing loss than normal age-related effects,” says Eva Karltorp, researcher at the Department of Clinical Science, Intervention and Technology at Karolinska Institutet and physician at the CI section at Karolinska University Hospital in Huddinge.
In practice, however, there are some age restrictions. For the procedure to be successful, the person receiving a CI implant must be willing and able to train in order to understand the sounds, which may initially sound different than usual. Surgery with anaesthesia can also be stressful for the body, especially for older people. There is also some risk of impacting the balance organ, which is also located in the ear, and this can increase the risk of falls in older people.
And then there must be functioning cochlear nerves. The cochlear implant does the job of the hair cells, so the damage must be there.
The technology captures sound with a microphone and sends it to a receiver located under the skin near the ear. The receiver has a connecting electrode, a thin cord, which extends into the cochlea. There it can stimulate the nerve fibres. Then sound waves reach the auditory centre of the brain. And that is where the sensory impression emerges.
But the final stage, the brain's ability to interpret, has an age-sensitive aspect. Those born into a silent world do not train their auditory centre. And then crucial functions for interpreting sensory input are irretrievably lost.
“We can help those who used to hear or young children born with deafness. We cannot help those who are born deaf and are older than perhaps four or five years. In such cases the result is not good,” says Eva Karltorp.
 Photo: privat
Photo: privatIda Bäckström suddenly lost her hearing
Ida Bäckström suddenly lost her hearing - first in the right ear and then in the left. Now she hears with the help of cochlear implants.
The younger the children are, the better the results.
“The age of surgery is the most important factor when it comes to deaf babies. But there are lots of other important factors, such as how much you use the implants and how much you are talked to, which differs between families and preschools. But we also think that it may matter what the nerve bundle looks like, how thick it is,” says Eva Karltorp.
She and her team are now studying, among other things, the language abilities of teenagers born deaf who received CIs as children. One study has already shown that children born deaf who had surgery before the age of nine months have similar language development to children born hearing, measured at the age of four.
Follow-up is now done on children who have had early surgery and have reached adolescence. Researchers look at perceived quality of life, grades in school and how well it is going with friends. They also conduct demanding spoken language tests that examine, for example, the understanding of non-literal phrases and figurative language. The results have not yet been published.
Previous studies have shown that at the group level, children with CI have more difficulty repeating so-called non-words, nonsense words without meaning. For example, if someone says “hepe blird” or “blarf cule”, children with CI generally have more difficulty repeating the words. The importance of being able to repeat non-words is not fully understood, but it is still considered to be linked to language impairment.
“These children's hearing works differently than the hearing in children born able to hear. And we know that there are individual differences. One question to pursue is whether this is something that can be influenced. Can operations be carried out even earlier? Or could the nerves need some additional stimulation? We do not know that today,” says Eva Karltorp.
About 4,500 people in Sweden live with cochlear implants. About 1,000 of them are children. Eva Karltorp has operated on about a thousand patients, both children and adults.
“I met one of those I operated on as a baby the other day. He was now studying at university and followed all the lectures. You would never have noticed that this person had a hearing loss, there was nothing in the conversation to indicate that.”
Test your hearing
On the website horseltestaren.se you can test how you perceive “speech in noise”, conversations in an environment with other sounds. The test was developed by The Swedish Association of Hard of Hearing People in collaboration with researchers.
Related feature articles
 Photo: Getty Images
Photo: Getty ImagesThe substances that can affect your hearing
 Photo: Getty images,Getty Images/iStockphoto
Photo: Getty images,Getty Images/iStockphotoTricky treatment of one-sided hearing loss
 Photo: Joey Guidone
Photo: Joey Guidone